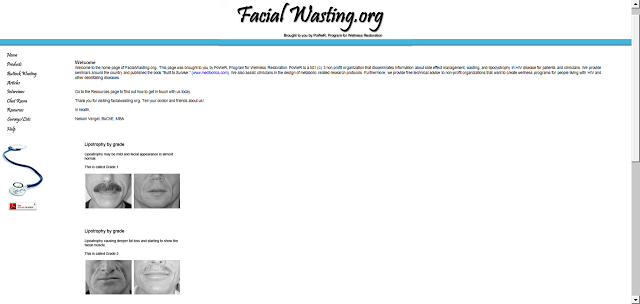
New Bill Mandates Coverage of HIV Lipodystrophy Treatments
Ben Klein, a Senior Attorney from GLAD.org speaks about the new bill approved in the state of Massachusetts in Aug. 2016 that mandates several insurance carriers in that state to reimburse cost for HIV related lipodystrophy therapies and how activists can advocate for similar bills in other states. California is also trying to get a […]
New Bill Mandates Coverage of HIV Lipodystrophy Treatments Read More »